Compounds of silicon8/11/2023 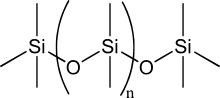
Please help improve this article by expanding this section with: continuous text and elimination of bullet points, giving better examples of silicon'schemistry and compounds and some citations. Silicon is a principal component of aerolites, which are a class of meteoroids, and also is a component of tektites, which are a natural form of glass. 
Asbestos, feldspar, clay, hornblende, and mica are a few of the many silicate minerals. These minerals occur in clay, sand and various types of rock such as granite and sandstone. Silicon also occurs as silicates (various minerals containing silicon, oxygen and one or another metal), for example feldspar. (They are known as "lithogenic", as opposed to " biogenic", silicas.) Sand, amethyst, agate, quartz, rock crystal, chalcedony, flint, jasper, and opal are some of the forms in which silicon dioxide appears. Silica occurs in minerals consisting of (practically) pure silicon dioxide in different crystalline forms. Silicon is usually found in the form of silicon dioxide (also known as silica), and silicate. Pure silicon crystals are only occasionally found in nature they can be found as inclusions with gold and in volcanic exhalations. Measured by mass, silicon makes up 25.7% of the Earth's crust and is the second most abundant element on Earth, after oxygen. It is similar to glass in that it is rather strong, very brittle, and prone to chipping. In its crystalline form, pure silicon has a gray color and a metallic luster. The electrical resistance of single crystal silicon significantly changes under the application of mechanical stress due to the piezoresistive effect. Pure silicon has a negative temperature coefficient of resistance, since the number of free charge carriers increases with temperature. Having four bonding electrons however gives it, like carbon, many opportunities to combine with other elements or compounds under the right circumstances.īoth silicon and carbon are semiconductors, readily either donating or sharing their four outer electrons allowing many different forms of chemical bonding. Even though it is a relatively inert element, silicon still reacts with halogens and dilute alkalis, but most acids (except for some hyper-reactive combinations of nitric acid and hydrofluoric acid) do not affect it. The outer electron orbitals (half filled subshell holding up to eight electrons) have the same structure as in carbon and the two elements are very similar chemically. It is much more important to the metabolism of plants, particularly many grasses, and silicic acid (a type of silica) forms the basis of the striking array of protective shells of the microscopic diatoms. Silicon is an essential element in biology, although only tiny traces of it appear to be required by animals. It is also a component of silicones, a class-name for various synthetic plastic substances made of silicon, oxygen, carbon and hydrogen, often confused with silicon itself. In the form of silica and silicates, silicon forms useful glasses, cements, and ceramics. Silicon is widely used in semiconductors because it remains a semiconductor at higher temperatures than the semiconductor germanium and because its native oxide is easily grown in a furnace and forms a better semiconductor/dielectric interface than almost all other material combinations. Elemental silicon is the principal component of most semiconductor devices, most importantly integrated circuits or microchips. On Earth, silicon is the second most abundant element (after oxygen) in the crust, making up 25.7% of the crust by mass. As the eighth most common element in the universe by mass, silicon occasionally occurs as the pure free element in nature, but is more widely distributed in dusts, planetoids and planets as various forms of silicon dioxide or silicate. A tetravalent metalloid, silicon is less reactive than its chemical analog carbon. Silicon ( pronounced /ˈsɪl ɪkən/ or /ˈsɪl ɪkɒn/, Latin: silicium) is the chemical element that has the symbol Si and atomic number 14. Dark grey with bluish tinge Standard atomic weightĤ, 3, 2, 1 (amphoteric oxide)
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
